FibermarX™
Radiopaque Tissue Marker
FDA-cleared FibermarX™ is a device intended to be implanted into the body to accurately visualize and constitute the reference frame for stereotactic radiosurgery and radiotherapy target localization. FibermarX™ is also used to mark tissues for future medical procedures such as intensity-modulated and image-guided radiation therapy (IMRT/IGRT).

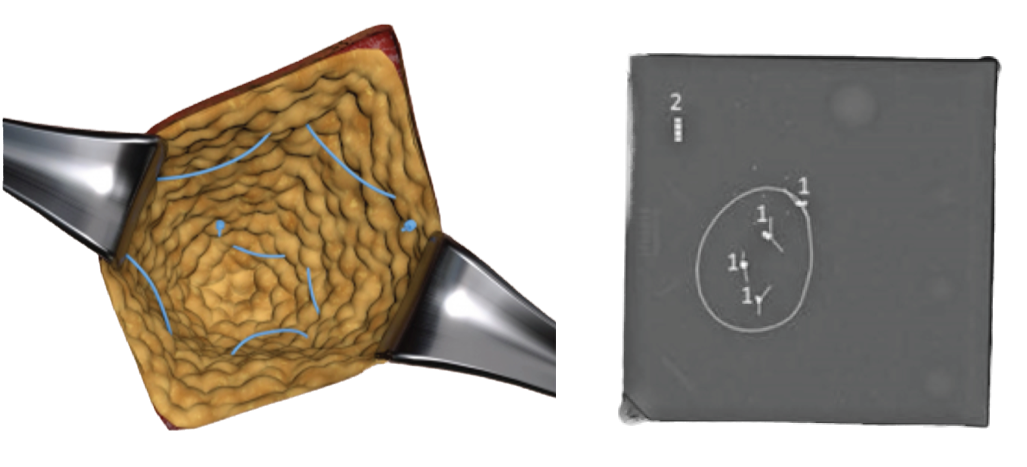

- Visible on CT, X-ray, or Mammography
- FDA Cleared
- EtO Sterilized
- 36” of Marker in Each Sterile Pouch
- Marker Denoted by White Arrows
FibermarX™ is a sterile, single-patient-use, barium sulfate infused non-absorbable polymer monofilament that is visible on standard radiographs (x-ray, CT, mammography). The device is passed through soft tissue and tied into place during open, percutaneous, or arthroscopic/laparoscopic/endoscopic procedures. Standard surgeon’s knots or a continuous running outline are used to quickly mark the soft tissue for subsequent imaging or for radiotherapy target localization.
FibermarX™ characteristics, proof-of-concept, and clinical use data are available in the open access journal paper published by Sahoo et al. in PLoS One (2019).


Extracellular Matrix
Biomaterials

Medical
Applications

Design
Services

Contract
Manufacturing
