XenoMEM™
Porcine Peritoneal Membrane
FDA-cleared XenoMEM™ Wound Matrix is a device indicated for the management of wounds. XenoMEM™ biologic scaffold materials are suitable for a host of regenerative medicine applications.


XenoMEM™ Structure and Composition
XenoMEM™ Wound Matrix is an acellular, porcine peritoneal matrix, supplied sterile to maintain and support an environment for wound management. It consists of an extracellular tissue matrix, derived from porcine peritoneum. XenoMEM™ Wound Matrix porcine peritoneal membrane provides a robust biological matrix that allows for easier handling during preparation and application of the wound dressing. The membrane has undergone a decellularization, viral inactivation and a freeze-drying process to remove donor genetic material in a non-destructive manner and maintain the structure and function of the tissue.
Composed of collagen type I, collagen type III, collagen type IV, fibronectin, elastin, laminin and other key ECM components, XenoMEM™ represents the ideal milieu for cell attachment and proliferation. In addition, the preservation of growth factors such as VEGF, FGF-b and TGF-b, grants XenoMEM™ with excellent cytocompatibility and biocompatibility properties, as supported by several studies published in peer-reviewed journals1-6.

XenoMEM’s composition preserves ECM components of the native peritoneum that play pivotal roles in cell attachment, migration and proliferation, with collagens making up to ~70% of total dry weight. Collagen type IV and laminin are the main components of basement membranes and are preserved on the XenoMEM™ basement membrane side and in blood vessels within the material. Also, a prominent network of elastin (~4% of dry weight) is preserved which provides the characteristic mechanical properties of the peritoneum. These ECM components are recognized by common hydrolytic enzymes found in wounds and during the normal healing process such as matrix metalloproteinases and elastases2, which results in the remodeling of XenoMEM™ upon implantation.
In addition, XenoMEM™ contains significant amounts of growth factors that enhance cell cytocompatibility and proliferation. These bioactive molecules include VEGF, FGF-b and TGF-b, which upon release during degradation promote proliferation of cells such as fibroblasts1.
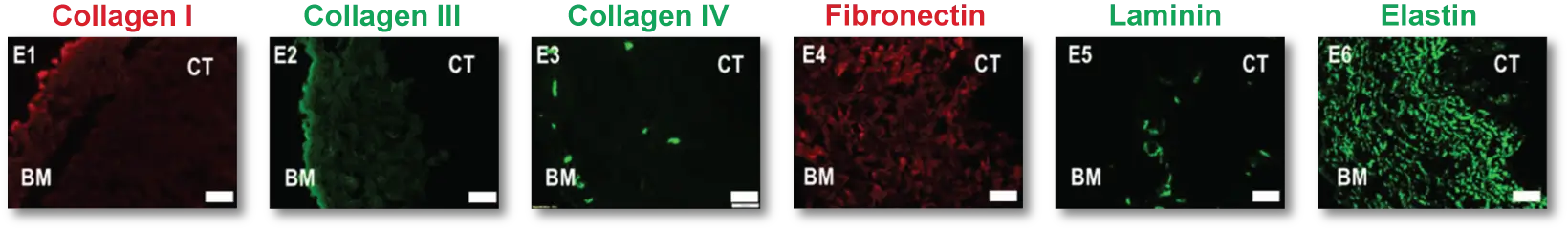

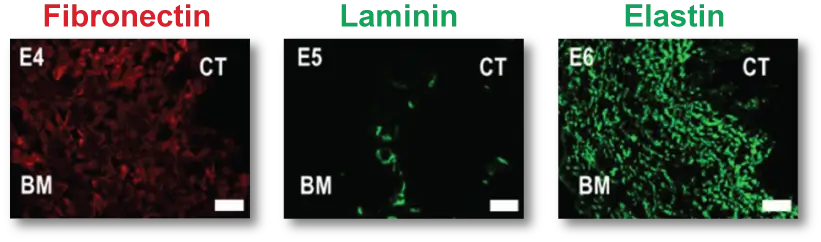
XenoMEM™ preserves critical components of ECM such as collagen and elastin, together with basement membrane components (collagen type IV and laminin).
An Elastic and Malleable Material
Due to the retention of ECM structure and integrity, XenoMEM™ provides strong and elastic mechanical properties, reaching a strength of ~30 MPa at break, and presenting a high resistance for suture retention1. In addition, it easily rehydrates within 5 minutes of incubation in saline at room temperature, absorbing up to two times its weight in water, which also allows the material to absorb exudates from the wound bed. XenoMEM™ is thermically stable up to 50 °C, preventing its denaturation upon implantation at body temperature.
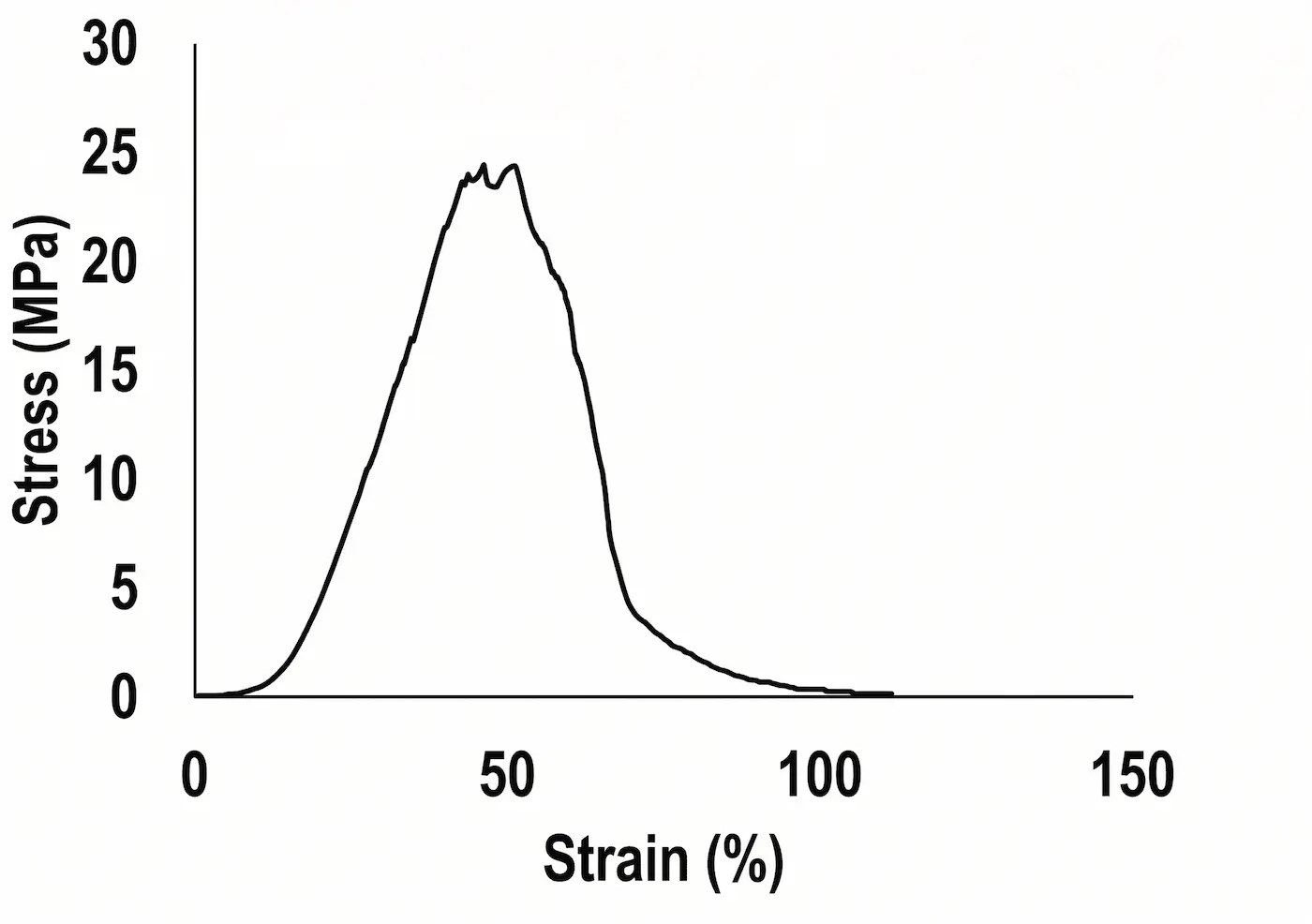
XenoMEM™ presents the typical strain-stress curve of native collagen-based tissues.
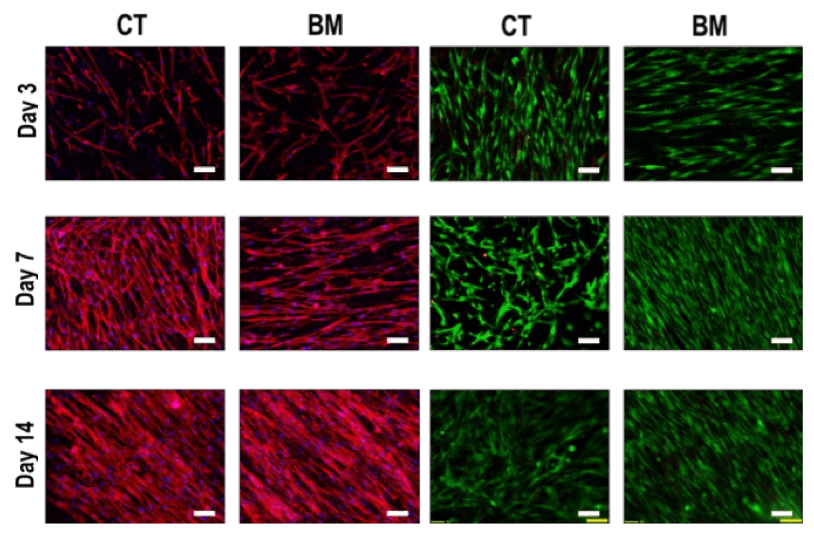
Human dermal fibroblasts attach and proliferate on both XenoMEM™ layers: basement membrane (BM) and connective tissue (CT), as shown by immunofluorescence of the cytoskeleton (red) and nuclei (blue) – left panel – and live cells (green) – right panel.
Cytocompatibility and Biocompatibility
XenoMEM™ compatibility with cells in vitro has been reported in several studies using cell lines, such as endothelial cells (HUVECs 2) or monocytes (THP-11,2), and primary cells, such as human dermal fibroblasts, human tenocytes1,2, human adipose derived stem cells3 and human mesothelial cells4. These findings represent the high cytocompatibility of XenoMEM™ that plays a key role in healing events such as angiogenesis, immune response, and ECM deposition. In addition, this material has also been shown to maintain the viability and biological activity of modeled exogenous tissues in ex vivo experiments such as rat aortic rings2. Overall, the ECM components and growth factors preserved within XenoMEM promote cell attachment and rapid cell proliferation. In addition, an absence of acute inflammatory reaction has been observed when seeding macrophages on XenoMEM™ confirming its minimal risk of immune response1,2.
Moreover, the basement membrane components and topography allow for a faster proliferation on the basal side of XenoMEM™, as observed with adipose derived stem cells, where stem cells maintained their multipotency and viability after 21 days of culture in vitro3. In HUVECs and rat aortic ring assays, XenoMEM™ proved its angiogenic properties mainly granted by the presence of soluble proangiogenic factors such as VEGF1,2.

Human dermal fibroblasts attach and proliferate on both XenoMEM™ layers: basement membrane (BM) and connective tissue (CT), as shown by immunofluorescence of the cytoskeleton (red) and nuclei (blue) – left panel – and live cells (green) – right panel.
Advanced Regenerative Medicine Applications
While XenoMEM™ has proven its efficacy in wound healing management, this material holds great potential for additional tissue engineering and regenerative medicine applications.
XenoMEM™ has been successfully tested as an adipose derived stem cells delivery system in a preclinical wound healing model. XenoMEM™ in combination with ADSCs promoted the healing of a full thickness wound while also enhancing new vessel formation3. The basement membrane side of XenoMEM™ boosted ADSCs proliferation and survival upon implantation, while maintaining the multipotency of the stem cells. This study proves the potential of XenoMEM™ for localized delivery of stem cells, a field that is in the forefront of regenerative medicine.
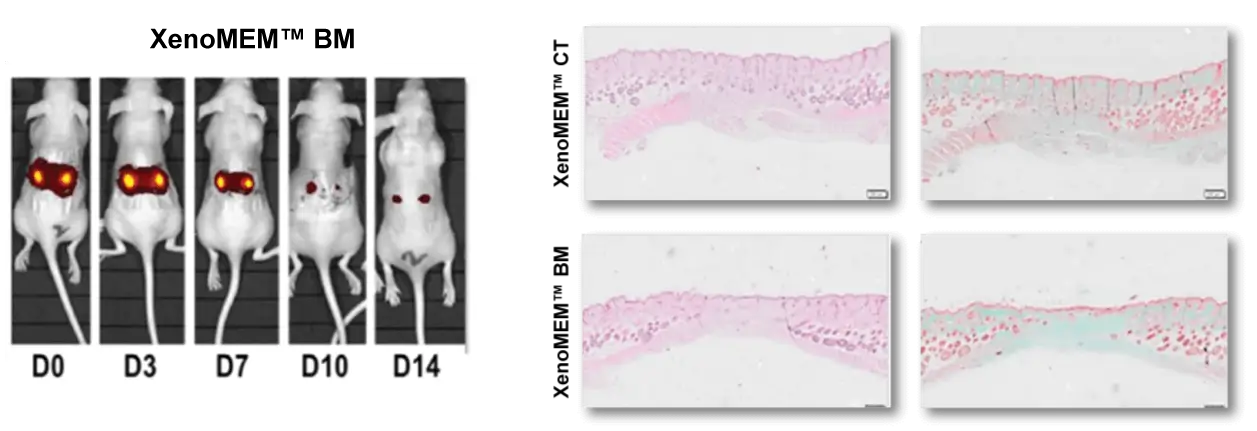
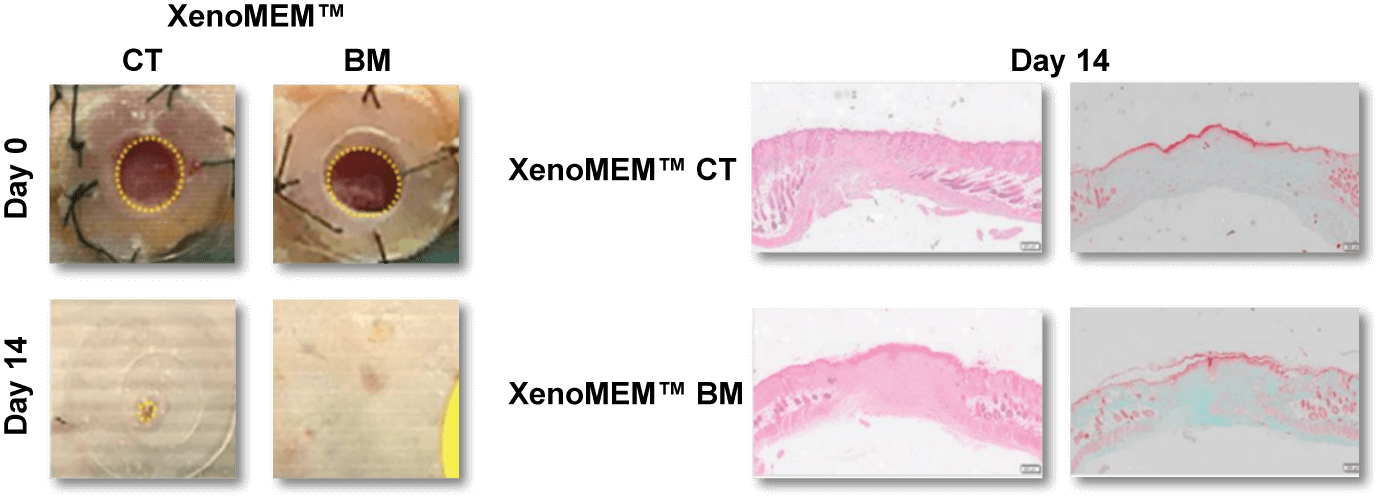
XenoMEM™ implanted in nude mice promotes wound healing and completely remodels.
The aforementioned applications have been demonstrated in XenoMEM™ formatted as 2D sheet. However, XenoMEM™ can be further form-factored to be used in other biomedical applications. As a micronized powder, XenoMEM™ has shown cytocompatibility and rapid invasion by dermal fibroblasts, providing a granular environment for cell proliferation. Moreover, solubilized XenoMEM™ can be used as a coating system, bioink, or hydrogel by providing a biologically active material.
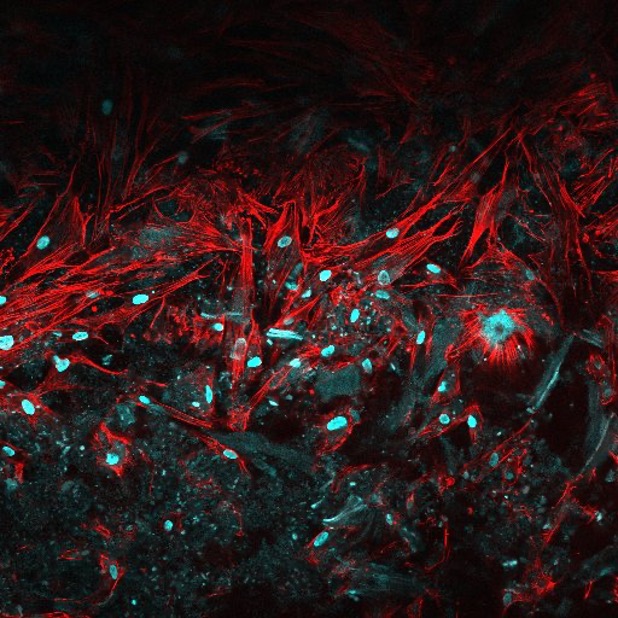
Micronized XenoMEM™ allows penetration and proliferation of fibroblasts after 7-day in vitro culture (red: cytoplasm, blue: nuclei).
References
1Capella-Monsonís, H., Kelly, J., Kearns, S., & Zeugolis, D. I. (2019). Decellularised porcine peritoneum as a tendon protector sheet. Biomedical Materials, 14(4), 044102.
2Capella-Monsonís, H., Tilbury, M. A., Wall, J. G., & Zeugolis, D. I. (2020). Porcine mesothelium matrix as a biomaterial for wound healing applications. Materials Today Bio, 7, 100057.
3Capella-Monsonís, H., De Pieri, A., Peixoto, R., Korntner, S., & Zeugolis, D. I. (2020). Extracellular matrix-based biomaterials as adipose-derived stem cell delivery vehicles in wound healing: a comparative study between a collagen scaffold and two xenografts. Stem Cell Research & Therapy, 11(1), 510.
4Capella-Monsonís, H., & Regenerative, M. (2020). Porcine peritoneum: A multifunctional xenograft for a diverse range of clinical indications (Doctoral dissertation, NUI Galway).
5Development of a Novel Nanoparticle Antimicrobial Dressing for the Prevention of Wound Infections. Interim report, University of Wisconsin – Madison.
6Alobaid, S.A., Shrestha, S., Tasseff, M. et al. Activity of silver-zinc nanozeolite-based antibiofilm wound dressings in an in vitro biofilm model and comparison with commercial dressings. Discover Nano 20, 26 (2025).

Extracellular Matrix
Biomaterials

Medical
Applications

Design
Services

Contract
Manufacturing