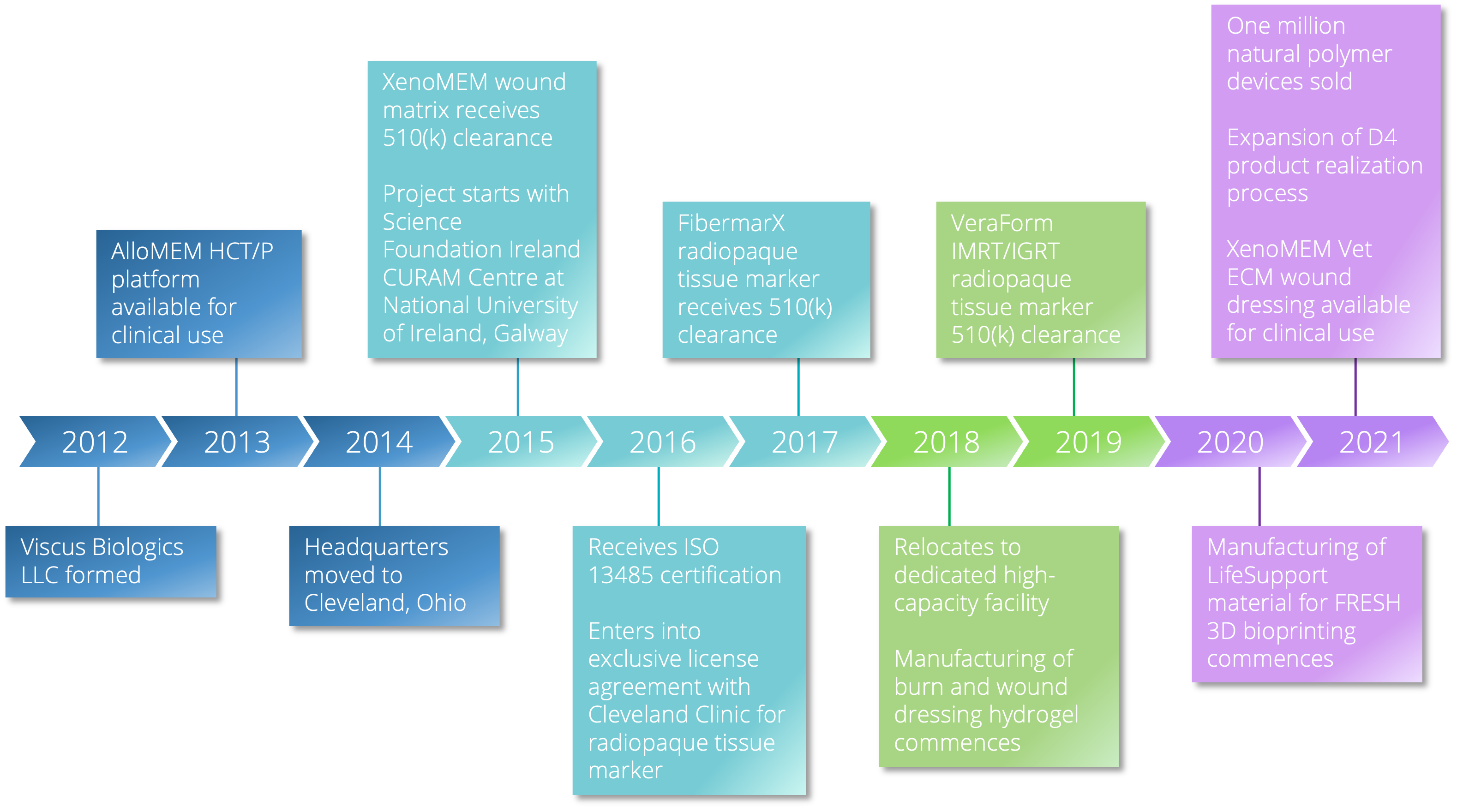
Viscus Biologics LLC specializes in helping people in the life science market segment using its established D4 process. Viscus Biologics is FDA registered with expertise in processing high performance biomaterials under a design services and contract manufacturing business model. Native polymers are processed into components, devices, and products. Several FDA cleared devices are produced in a controlled clean room setting.
Founded in 2012 to help companies solve complex challenges ranging from ideation to high volume production. We operate in centers of excellence for manufacturing and health care delivery.

World Headquarters
Cleveland, Ohio

Pre-Clinical Research
Dublin, Ireland
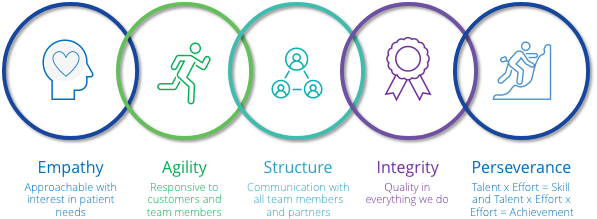
The culture at Viscus Biologics emanates from core values shared by team members throughout the organization.


Donna Campbell
Director of Quality


Nicole Fraifogl, PhD
Lead Project Engineer

Laura Schramm
Office Administrator
Viscus Biologics, LLC designs, manufactures and delivers innovative medical devices with the objective of positively impacting the health and well-being of patients around the world. Given the nature of these products, Quality is of paramount importance in the culture and operation of the organization.
Accordingly, Viscus Biologics, LLC has established a Quality Management System in compliance with ISO 13485, FDA 21 CFR 820 and other appropriate national regulations to guide our actions and ensure safe and effective devices. Viscus Biologics, LLC, is committed to establishing a quality culture and providing quality products by:





